 |
| Colon cancer cells: on the move and stuck in place. |
Moving cells make an excellent drug target. But what proteins should the drugs be aimed at? First researchers must learn how a tangled mesh of proteins keeps a cell motoring forwards.
|
|
|
A slice of fish skin or a chunk of cow heart in a culture dish doesn't sit there quietly. The cells march across the dish in a futile attempt to heal a perceived wound. The same mobilization happens in people: cells called fibroblasts flood into cuts, immune cells burrow through tissues to seek out invaders, embryonic cells clamber over each other to form young organs, and cancer cells spread. Our body seethes with movement.
Drugs that exploit subtle variations in cell movement might keep cancer cells stuck in one place while leaving immune cells free to roam. For that reason, the National Cancer Institute has investigated chemicals that trip up a key movement protein. And a company called Cytokinetics, Inc. (South San Francisco, California) is also keeping a close eye on cell movement research. Cytokinetics is interested in drugs that mess with the cell's internal system of structural beams and highways, called the cytoskeleton. "There's a richness of understanding of the role of the cytoskeleton," says Cytokinetics CEO James Sabry, "and that for us represented a real opportunity."
One of the cytoskeleton's most important jobs is to move cells around. Although Sabry anticipates tackling cell movement in the future, for now he is focussing on other functions of the cytoskeleton, such as cell division. What Sabry is waiting for is a better understanding of how the cell movement machine works. For such an important process, an understanding of the mechanics of cell movement has remained surprisingly obscure.
But that picture is changing. In recent work, scientists have looked at the location and movements of single proteins, and managed to convert a disorganized parts list into a blueprint for a working engine. There are no pistons or flywheels in this engine, just a dense protein mesh that somehow creates movement.
|
|
|
Any design for the engine has to account for one fact: the engine is in a constant cycle of decay and rebirth. The cell walks over its own engine, most of which remains stationary, even as the cell creates a new version of the engine at the cell front. The researchers' new blueprints for the engine have a natural beauty, because the mechanism for constantly recreating the engine is a logical byproduct of the basic motor design.
The motor's other trick is that it must produce asymmetry where once there was none. Your average cell is like a balloon filled with jelly, but somehow this balloon can transform itself into a movement machine. It's as if the parts of a car engine, initially strewn across a floor, could grab onto each other and take the car out for a spin. Suddenly the cell has a front that pushes outwards and attaches to its surroundings, a middle that pulls the rest of the cell forwards with a molecular motor, and a back that lets go.
Some cell movement researchers invoke complexities worthy of a jet fighter engine, but Gary Borisy of the University of Wisconsin, Madison, prefers the image of a two-stroke lawnmower. Borisy is the movement minimalist - in his world two proteins called actin and myosin can explain just about everything.
But first he had to show that they were even relevant.
Actin and myosin are logical suspects for a movement motor. These two proteins are at the front of moving cells, and they are already known to produce force: in muscle the glob-like myosin motor protein grabs onto cables of actin and slides them over each other. The increased cable overlap makes a muscle cell collapse in on itself, or contract, and it can help a compartmentalized human body move around. But contraction alone, for a single cell, is useless. It makes a cell shorter and fatter, but it won't get it from one place to another.
Myosin suffered another blow to its credibility when William Loomis of the University of California, San Diego, and James Spudich of Stanford University (Palo Alto, California) got rid of it in a crawling amoeba and found that the cells could still move. "Up until those papers the contractile models for cell locomotion were widely discussed," says Tim Mitchison of Harvard Medical School in Boston. "Then it became dogma in the field that movement doesn't involve myosin II."
|
|
|
If myosin wasn't the culprit, perhaps actin was. The extension of actin cables is known to push out the front of the cell, and with a strong enough push the rest of the cell might be dragged along behind. A food-poisoning bug called Listeria uses a similarly simple system: it hijacks the cell's actin to make a propulsive tail - no myosin necessary. Worm sperm, meanwhile, clearly don't use myosin. They don't have any actin either, but they use a functionally similar cable to push out the front of the cell.
But Borisy still had faith in myosin, especially as researchers began to identify other versions of myosin that could have been filling in for the one removed by Loomis and Spudich. Now Borisy just had to find the myosin amongst the tangled cables of actin at the front of the cell, and work out how such an unholy mess was driving cell movement. The labor was hard but worthwhile. "This is absolutely beautiful work," says Mitchison. "It has rehabilitated myosin."
|
|
|
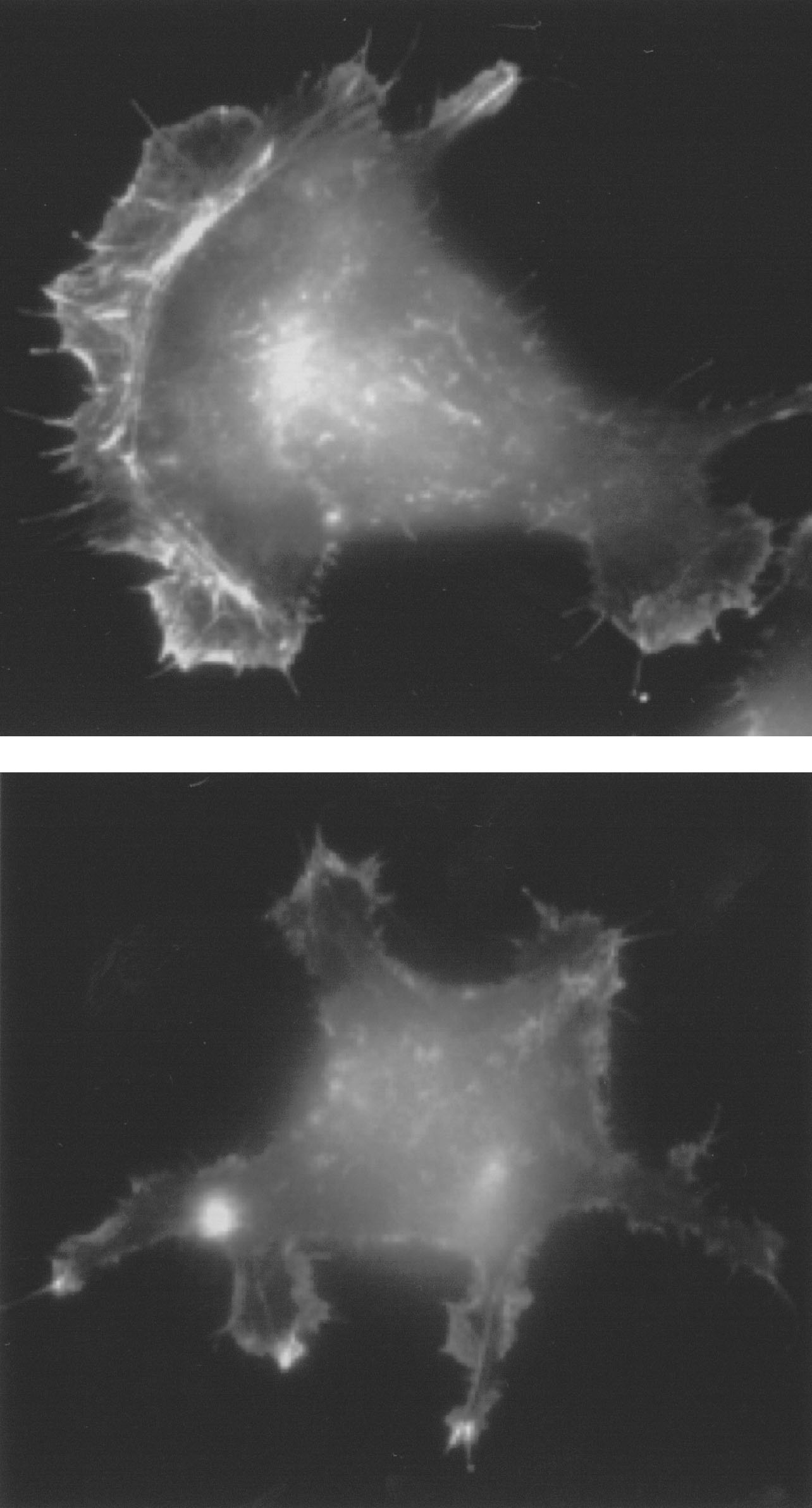
Borisy chose to study fish skin cells — the masters of movement. In an hour they cover just under a millimeter, which on a larger scale is roughly equivalent to a person-sized bag of goo sloshing from one end of a football field to another. The cut-healing fibroblasts in humans are larger and around ten times slower ("like studying flight in an ostrich," according to one researcher); perhaps, speculates Borisy, "fish are wounded more than we are."
The fish skin cells are so fast that they can be made into headless chickens. Borisy reported early in 1999 that he could make hundreds of these cell fragments by adding a chemical that turns off myosin. Actin cables kept growing and pushing out on the front of the cell. But without myosin dragging the rest of the cell forwards, the majority of the cell could not keep up. When the tension became too much the front of the cell broke away, motoring across the culture dish. It left behind the nucleus - the command-central of the cell. But still the headless-chicken cells kept going.
The headless chickens prove that there is an independent motor in the front of the cell. These cell fragments don't need building materials from the nucleus, or large actin roadways running across the whole cell, or signals from outside the actin network telling it which way is forwards. "The movement machinery, once polarized, propagates its own polarized state," says Borisy. The fragments' remarkable self-sufficiency, coupled with their small size and simplicity, mean they are a great place to start understanding how movement is generated.
|
|
|
Borisy deciphered the motor's design by watching its creation. The ignition switch was a gentle puff of fluid on some stationary fragments to coax them into movement. The push squished together enough myosin to start a contraction, thus lifting what was now the 'back' of the cell. The cell then acted like a plastic paddling pool, in which a push on one side makes water slop into the bulge on the other side. The only actin that could grow into the new space on the other side was the actin that was already pointing forwards, so this actin pushed the cell edge further outwards and forwards. The changes were self-perpetuating - actin pushing at the front made the cell rear bunch up; the resulting myosin contraction pulled up the back, making more room for actin extension at the front - and the cell fragment sped off.
The same two-part motor seems to work in whole fish cells. Borisy found that strong actin cables push out the very front of the cells, but, further back from the front, the cables weaken and can be deformed by groups of myosin motors. The myosins run along multiple splayed actin cables at once and act like a zipper, collapsing the loose mesh into a tight bundle. The mesh is anchored to the surface below the front of the cell, so it collapses forwards, and connecting cables running back to the nucleus pull everything else forward with it.
|
|
|
Borisy's model predicts that a moving cell will exert force near where the mesh is collapsing. Sure enough, when Michael Sheetz of Duke University watches cells move over a series of tiny cantilevers, the cells exert the greatest forces on the cantilevers that are underneath the collapsing mesh.
Case closed, at least for fish skin. In fibroblasts Mitchison and Louise Cramer of University College, London, think that myosin may drag the nucleus forward on large, established roadways of actin, although Borisy says no one has shown that those roadways are relevant to movement.
But neither Borisy nor Cramer claim that two proteins are enough for all cells. Larger cells need something more, and, like actin and myosin, that something must be capable of recreating itself as the cell moves forwards.
For the sluggish fibroblasts, the extra ingredient is a set of train tracks called microtubules - thin tubes that radiate from the front of the nucleus like the rays of the sun. Microtubule tracks are known as highways for delivering building materials, so it had been presumed that they were fulfilling the same function at the front of moving cells. But fibroblasts, when they are poisoned with anti-cancer drugs that eliminate microtubules, seem to be lacking not bricks and mortar but organization. "They attempt to go in all directions simultaneously," says Borisy. "The rationalization is that with a large cell you need some way to control all the pieces," so perhaps the microtubules are the master conductor that defines front and back. "That's very far from molecules or mechanisms, but that's where the field is," says Borisy. "It's a frontier area."
|
|
|
The first clue as to how microtubules work came in May 1999 from Ted Salmon of the University of North Carolina at Chapel Hill. He found that microtubules deliver a signal that makes the front of the cell poke out and then attach. But then Victor Small of the Austrian Academy of Sciences in Salzburg reported a contradictory ungluing effect of microtubules in a September 1999 article. Small watched microtubules as they lengthened and shortened, and found that they were acting like smart bombs. An extraordinary number of them were lured exactly to the sites where actin cables are anchored to the surface underlying the cell. Sites at the back of the cell were targeted eight times more frequently than those at the front, and arrival of a microtubule was often followed soon after by disappearance of the anchor.
The adhesion-dissolving may be needed to compensate for the fibroblasts' indecision. "The contacts at the back are mostly formed when the cell was thinking of going in that direction but then changed its mind," says Small. The microtubules should help the back to unstick itself and keep moving forwards.
|
|
|
If Salmon and Small are both correct, microtubules must be delivering different signals to the front and back of the cell. Small doesn't feel the need to explain this situation because, he says, "we think they're wrong." But Salmon has a possible solution. He has seen microtubules get swept from the front of the cell by the backwards flow of excess, newly formed actin. The microtubules are bent and broken by the flow, and the exposed ends may load up with different cargoes to deliver to the back of the cell. Thus the actin controls microtubules, even as microtubules control actin. This self-sustaining cycle is layered on top of Borisy's basic cycle of reciprocal actin and myosin control.
|
|
|
The cycles are so strong that the cellular engine never stops - it just idles. In the November 5 1999 issue of Science, Eugene Marcantonio and Gregg Gundersen of Columbia University in New York took the unusual step of looking at stationary fibroblasts to see what, if anything, the cells were doing. To their surprise, the cells were slowly forming and then pulling in their actin anchor points. The cells are stuck in place "not because their feet are stuck down too hard," says Marcantonio, but because they are waiting for a signal to put them into gear. "We propose," he says, "that there is a molecular clutch that engages the movement."
Identifying the clutch and the messages delivered by microtubules will be important future steps, but not the last. Cataloging parts is only a start in understanding a motor that constantly changes over time and space. "How will you know that you have solved the problem?" asks Mitchison. "It may have to be solved by computer modeling. Success will be when you can put the parameters into a computer, and get your model cell to walk."